  
Logunov DY, Dolzhikova IV, Shcheblyakov DV, et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. The authors report no other conflicts of interest in this work. Ministerio de Ciencia, Tecnología e Innovación, Argentina Freely provided drug and placebo samples from Laboratorio Pablo Cassará, during the conduct of the study personal fees from Laboratorio Pablo Cassará, outside the submitted work Dr Mónica Lombardo report personal fees from Laboratorio Pablo Cassará, outside the submitted work, is a Director of Scientific Affairs at Nobeltri S.R.L Dr Robert Giugliano report personal fees from Astra Zeneca, personal fees from CVS Caremark, personal fees from Pfizer, personal fees from Novartis, outside the submitted work and Institutional research grant to the TIMI Study Group at Brigham and Women’s Hospital for research he is not directly involved in from Abbott Aralez AstraZeneca Bayer Eisai GlaxoSmithKline Intarcia Janssen Research and Development Medicines Company MedImmune Novartis Poxel Pfizer Quark Pharmaceuticals Roche Takeda Zora Biosciences. In this pilot study a nasal spray with I-C showed significant efficacy in preventing COVID-19 in health care workers managing patients with COVID-19 disease.ĬOVID-19 hospital workers nasal prophylaxis spray.ĭr Juan Manuel Figueroa report grantfrom Programa de articulación y fortalecimiento federal de las capacidades en ciencia y tecnología COVID-19, Proyecto CABA 20. Absolute risk reduction: 4% (95% CI 0.6 to 7.4). The incidence of COVID-19 differs significantly between subjects receiving the nasal spray with I-C (2 of 196 ) and those receiving placebo (10 of 198 ). Both treatment groups had similar baseline characteristics. The trial is registered at ( NCT04521322).Ī total of 394 individuals were randomly assigned to receive I-C or placebo. The primary end point was clinical COVID-19, as confirmed by reverse transcriptase polymerase chain reaction testing, over a period of 21 days. Clinically healthy physicians, nurses, kinesiologists and other health care providers managing patients hospitalized for COVID-19 were assigned in a 1:1 ratio to receive four daily doses of I-C spray or placebo for 21 days. This is a pilot pragmatic multicenter, randomized, double-blind, placebo-controlled study assessing the use of a nasal spray containing I-C in the prophylaxis of COVID-19 in hospital personnel dedicated to care of COVID-19 patients. In vitro, I-C inhibits SARS-CoV-2 infection in cell culture.Ĭan a nasal spray with Iota-Carrageenan be useful in the prophylaxis of COVID-19 in health care workers managing patients with COVID-19 disease? Iota-Carrageenan (I-C) is a sulfate polysaccharide synthesized by red algae, with demonstrated antiviral activity and clinical efficacy as nasal spray in the treatment of common cold. 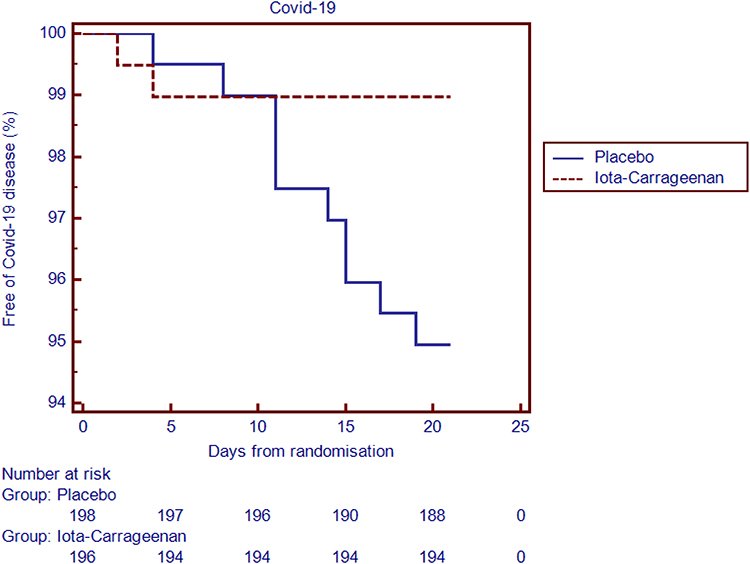
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
